Lab testsBlood samples can be checked for levels of the enzyme associated with Gaucher disease. Genetic analysis can reveal whether you have the disease. Imaging TestsPeople diagnosed with Gaucher disease typically require periodic tests to track its progression, including:. Dual energy X-ray absorptiometry (DXA).
Type 1 Gaucher's disease is frequent among Jewish people of Eastern European origin; the carrier frequency in these individuals is approximately 1 in 15, whereas the disease frequency is as high as 1 in 855. Many type 1 affected individuals never come to medical attention, contributing to an underestimation of frequency.
This test uses low-level X-rays to measure bone density. MRI. Using radio waves and a strong magnetic field, an MRI can show whether the spleen or liver is enlarged and if bone marrow has been affected.Preconception screening and prenatal testingYou might want to consider genetic screening before starting a family if you or your partner is of Ashkenazi Jewish heritage or if either of you have a family history of Gaucher disease. In some cases, doctors recommend prenatal testing to see if the fetus is at risk of Gaucher disease.
More Information.TreatmentWhile there's no cure for Gaucher disease, a variety of treatments can help control symptoms, prevent irreversible damage and improve quality of life. Some people have such mild symptoms that they don't need treatment.Your doctor likely will recommend routine monitoring to watch for disease progression and complications. How often you'll need to be monitored will depend on your situation. MedicationsMany people who have Gaucher disease have seen improvements in their symptoms after beginning treatment with:. Enzyme replacement therapy.
This approach replaces the deficient enzyme with artificial ones. These replacement enzymes are given in an outpatient procedure through a vein (intravenously), typically in high doses at two-week intervals. Occasionally people have an allergic or hypersensitivity reaction to enzyme treatment. Miglustat (Zavesca). This oral medication appears to interfere with the production of fatty substances that build up in people with Gaucher disease. Diarrhea and weight loss are common side effects. Eliglustat (Cerdelga).
Approved by the Food and Drug Administration in 2014 for treating the most common form of Gaucher disease, this drug also seems to inhibit the production of fatty substances that build up in people with this this condition.Possible side effects include fatigue, headache, nausea and diarrhea. Osteoporosis drugs. These types of medication can help rebuild bone weakened by Gaucher disease.Surgical and other proceduresIf your symptoms are severe and you're not a candidate for less invasive treatments, your doctor might suggest:. Bone marrow transplant. In this procedure, blood-forming cells that have been damaged by Gaucher disease are removed and replaced, which can reverse many of Gaucher signs and symptoms. Because this is a high-risk approach, it's performed less often than is enzyme replacement therapy.
Spleen removal. Before enzyme replacement therapy became available, removing the spleen was a common treatment for Gaucher disease. Now this procedure typically is used as a last resort. Coping and supportHaving any chronic illness can be difficult, but having a rare disease like Gaucher can be even harder.
Few people know about the disease, and even fewer understand the challenges you face. You might find it helpful to talk to someone else who has the Gaucher disease or someone who has a child with the disease. Talk to your doctor about support groups in your area. Preparing for your appointmentYou're likely to start be seeing your primary care provider.
Then you might be referred to a doctor who specializes in blood disorders (hematologist) or to a doctor who specializes in inherited disorders (geneticist). Gaucher disease: Pathogenesis, clinical manifestations and diagnosis. Accessed Jan.
6, 2017. Gaucher disease (Type 1). Genetic Disease Foundation. Accessed Jan. 6, 2017. Hughes D. Gaucher disease: Initial assessment, monitoring and clinical course.
Accessed Jan. 6, 2017. Gaucher disease. Merck Manual Professional Version. Accessed Jan. 6, 2017.
National Library of Medicine. Gaucher disease. Genetics Home Reference. Accessed Jan. 6, 2017. Hughes D.
Gaucher disease: Treatment. Accessed Jan. 6, 2017. FDA approves new drug to treat a form of Gaucher disease. Food and Drug Administration. Accessed Jan.
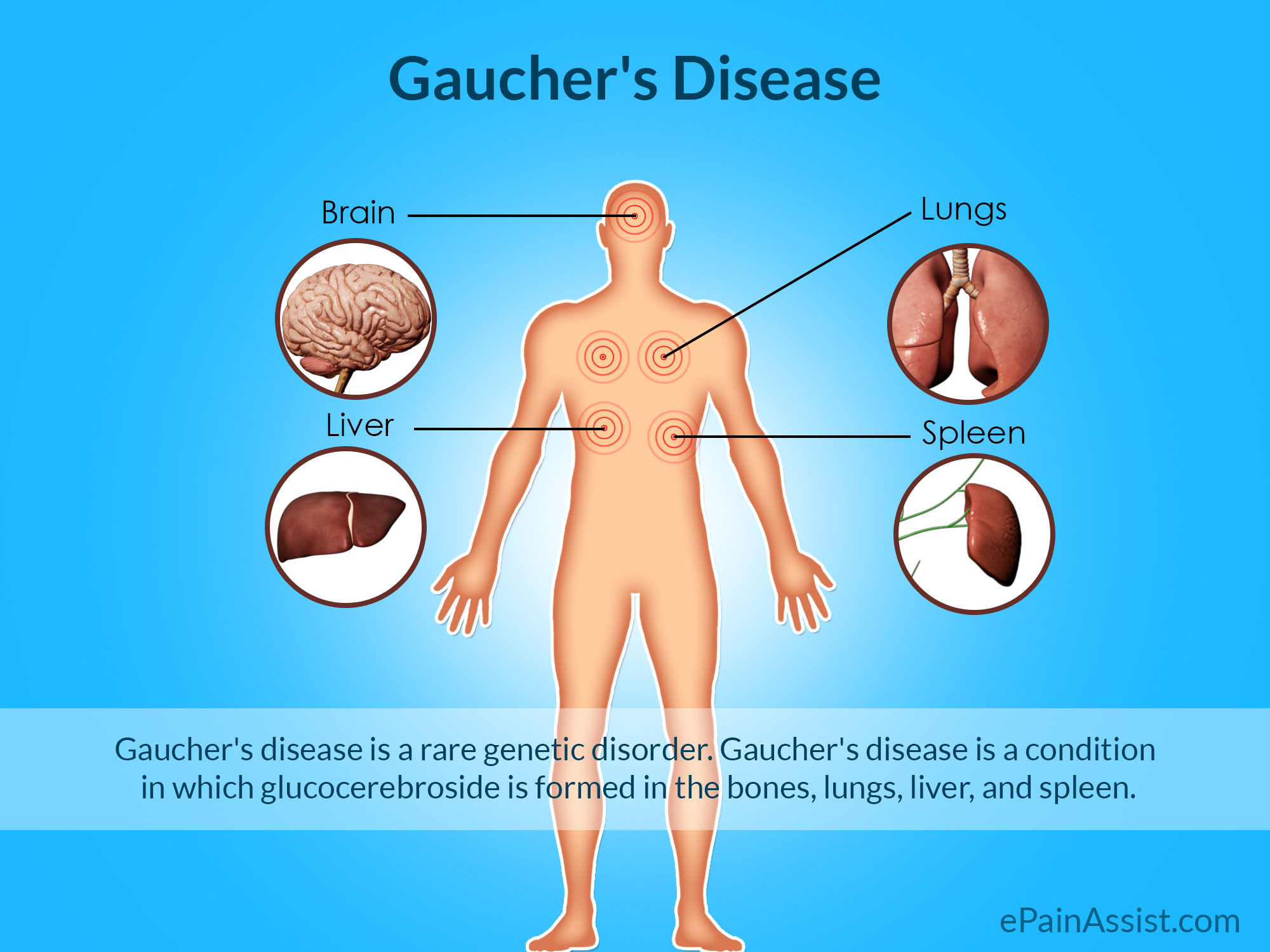
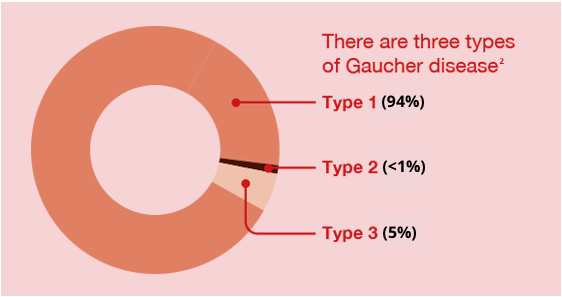
Synonyms: glucocerebrosidase deficiency, acid beta-glucosidase deficiencyGaucher's disease is the most common lysosomal storage disease and is caused by insufficient activity of the lysosomal enzyme acid beta-glucosidase (glucocerebrosidase), leading to the deposition of glucocerebroside in cells of the macrophage-monocyte system. Deficiency of acid beta-glucocerebrosidase causes widespread accumulation of glucosylceramide-laden macrophages. Glucosylceramide accumulation is widespread, including the bone marrow, liver, spleen and lungs. Central nervous system (CNS) involvement only occurs in patients with disease type 2 (acute neuronopathic) and type 3 (chronic neuronopathic) Gaucher's disease. There are three clinical subtypes:.
Type 1 - adult or non-neuronopathic form:. Often presents in childhood with hepatosplenomegaly, pancytopenia and skeletal disease. The severity of type 1 Gaucher's disease is extremely variable; some patients present in childhood with virtually all the complications of Gaucher's disease, while others are asymptomatic into the eighth decade. Patients diagnosed in the first five years of life are frequently non-Jewish and typically have a more malignant disease course. Type 2 (rare) - infantile form (acute neuronopathic).
This causes rapidly progressive neurovisceral storage disease and death during infancy. Type 3 - juvenile or Norrbottnian form (chronic or subacute neuronopathic).
This is less rapidly progressive neurovisceral storage disease, causing death in childhood or early adulthood. Epidemiology.
All three subtypes are inherited as autosomal recessive traits. The overall frequency of Gaucher's disease variants is about 1 in 40,000 to 1 in 50,000 live births. Type 1 Gaucher's disease is frequent among Jewish people of Eastern European origin; the carrier frequency in these individuals is approximately 1 in 15, whereas the disease frequency is as high as 1 in 855.
Many type 1 affected individuals never come to medical attention, contributing to an underestimation of frequency. Neuronopathic forms (types 2 and 3) are the rarest variants of Gaucher's disease, with an estimated incidence of fewer than 1 in 100,000 live births. Presentation Type 1 Gaucher's disease. May present with chronic fatigue, hepatomegaly, splenomegaly (may become massive), and bone involvement (bone pain due to bone infarcts or pathological fractures due to osteopenia) and may bruise easily or present with nosebleeds, bruising and petechiae (because of thrombocytopenia). Short stature and wasting occasionally are found in patients with massive organomegaly.
Occasionally present with pulmonary infiltration or portal hypertension.Type 2 Gaucher's disease. Presents in infancy with increased tone, strabismus, and organomegaly. Failure to thrive and stridor (due to laryngospasm) are also common. Rapid neurodegenerative course with extensive visceral involvement and death (usually caused by respiratory problems) within the first two years of life.
Type 3 Gaucher's disease. Presents in infancy or childhood. In addition to organomegaly and bony involvement, neurological involvement is present, including developmental delay and abnormal neurological findings - eg, increased tendon reflexes. It has been further classified as type 3a (with progressive myotonia and dementia) and type 3b (with isolated supranuclear gaze palsy) based on the extent of neurological involvement.Differential diagnosis. Any other cause of hepatosplenomegaly, thrombocytopenia, bone pain and osteopenia (type 1).
Any other cause of neurodevelopmental delay (types 2 and 3). Investigations General assessment. FBC and differential (assess the degree of pancytopenia); LFTs (minor elevations of liver enzymes are common but jaundice is a poor prognostic indicator). Skeletal radiography can detect and evaluate skeletal manifestations of Gaucher's disease. CXR to evaluate pulmonary manifestations.
Ultrasonography of the abdomen: determine extent of organomegaly. MRI scanning is more accurate in determining organ size and involvement. Patients with neuronopathic forms also need MRI scan of the brain, electroencephalogram (EEG) and diagnostic brainstem-evoked responses. Dual-energy X-ray absorptiometry (DEXA) scanning: evaluation of osteopenia.Diagnosis. Acid beta-glucosidase activity: this can be measured in peripheral blood leukocytes or cultured skin fibroblasts to confirm diagnosis. Heterozygotes have half-normal enzyme activity but there is an overlap with non-affected controls.
Acid beta-glucosidase genotyping: molecular diagnosis can be helpful, especially in Ashkenazi patients, in whom four mutations (N370S, 84GG, L444P, IVS2+1) in the acid beta-glucosidase gene account for nearly 97% of disease alleles. Bone marrow aspiration: diagnosis may be suggested by the finding of classic glycolipid-laden macrophages (Gaucher cells). This is not now the initial diagnostic test, as the blood enzyme test is sensitive, specific, and much less invasive. Prenatal diagnosis. There is carrier-screening for individuals of Ashkenazi Jewish descent to identify couples at risk of having a child affected with Gaucher's disease.Management , Management options include enzyme replacement therapy (ERT) and substrate reduction therapy (SRT), although bone marrow transplantation and gene therapy have been used (in rare cases). Management also includes treatment of complications such as anaemia, thrombocytopenia, bleeding tendency, skeletal disease, liver or lung involvement and organomegaly.Three different human recombinant enzymes are available: imiglucerase, velaglucerase alfa and taliglucerase alfa. Improvement in bone marrow and in the osseous skeleton in response to ERT occurs more slowly than the visceral and haematological responses.
Increase of bone mineral density in response to ERT can take up to eight years. Pathological damage such as osteonecrosis, bone infarctions and fractures are clearly irreversible. Bisphosphonates can be effective to increase bone density and prevent complications. Supportive management for bone pains or bone crises is frequently required and orthopaedic surgery may be needed for pathological fractures or osteonecrosis.Substrate synthesis inhibition therapy is an alternative approach, based on reduced synthesis of glucosylceramide by inhibiting the appropriate synthetic enzyme (glucosylceramide synthase).
Miglustat was approved in 2002 for patients with mild-to-moderate Gaucher's disease. However, adverse effects related to the use of miglustat limited its acceptance. Eliglustat is a new agent with a better safety profile and higher potency than miglustat.Bone marrow transplantation may be an effective treatment for neurological progression in this disorder.
However, there is significant morbidity and mortality and therefore it is not currently recommended in the management of neuronopathic Gaucher's disease. Gene therapy may offer the possibility of definitive therapy in the future.; Phenotype, diagnosis, and treatment of Gaucher's disease.
2008 Oct 4372(9645):1263-71.; Online Mendelian Inheritance in Man (OMIM).; Online Mendelian Inheritance in Man (OMIM).; Online Mendelian Inheritance in Man (OMIM).; Epidemiology of lysosomal storage diseases: an overview. 2006.; Clinical manifestations and management of Gaucher disease. Clin Cases Miner Bone Metab. 2015 May-Aug12(2):157-64.
Doi: 10.11138/ccmbm/2015.12.2.157. Epub 2015 Oct 26.; Gaucher disease: a lysosomal neurodegenerative disorder.
Eur Rev Med Pharmacol Sci. 2015 Apr19(7):1219-26.; Gaucher disease.
J Clin Exp Hepatol. 2014 Mar4(1):37-50. Doi: 10.1016/j.jceh.2014.02.005. Epub 2014 Apr 21.
